
At extremely high concentrations, the measured diffusion coefficient would not stem from self-diffusion as such, although it can be measured, the size calculated would not be the actual particle size. By lessening these interparticle interactions, the impact of mutual diffusion is lessened and as such, the mutual diffusion coefficient methods the value projected for self-diffusion as concentration is lessened. By producing serial dilutions of a concentrated solution or suspension, you gradually dilute away interactions across particles. The idea of infinite dilution is an abstraction it is not possible to measure an actual sample at a theoretical limit. This becomes increasingly complicated when interactions with other particles are measured. This is frequently not the case and crucial exceptions are outlined below. For a given q value, Г is associated with the translational diffusion coefficient, D T, through the following expression:Įmploying this calculation, all decays are regarded as stemming entirely from diffusion and all diffusion coefficients are regarded as though they stem from the thermal motion of a lone particle. 
The level of diffusion of the particle directly relates to the calculated size, where fast decay rates relate to smaller particles and lower decay rates relate to larger particles. For most compact globular particles, a single scattering angle can be employed to achieve hydrodynamic size. However, it is very common to measure at a single q, as is true with fixed-angle DLS measurements. The diffusion coefficient would arise from the slope of the above plot. The quickest decay relates to the smallest efficient particle size, which in the dilute case relates to the diffusion coefficient of a lone particle. Also of note, that flexible polymers frequently have multiple modes of relaxation corresponding to diffusion and also of internal motion of the polymer chain. Image Credit: Brookhaven Instrument Corporation Given two spherical particles, the larger particle will diffuse more slowly. The relationship between Г, and hydrodynamic size will be addressed in the following sections. Image Credit: Brookhaven Instrument Corporationįigure 1.1. Simulated ACF’s for decay rates of Г = 7000, 40 s -1 (corresponding to 24, 41 and 165 nm diameter spherical particles). This Г is the reciprocal of the characteristic relaxation time, τ t, such that:įigure 1.0. In the simplest instance, a single decay rate, Г, is extracted from the DLS autocorrelation function (ACF). This single particle diffusion coefficient is the outcome of self-diffusion as such, in the limit of infinite dilution, this calculated size is entirely equivalent to its hydrodynamic size. A lone particle would experience Brownian Diffusion in inverse proportion to its size as such, the diffusion coefficient, D T, of this article can be employed to work out its hydrodynamic diameter, d H, which can be seen in the Stokes-Einstein equation:ĭ H can be achieved if the temperature, T and viscosity, η, are also established. All freely diffusing particles are continuously experiencing Brownian Motion in proportion to thermal energy stated as the product of the Boltzmann constant by the temperature (k bT). The size acquired from DLS is a hydrodynamic size, which comes from measurements of particle diffusion. 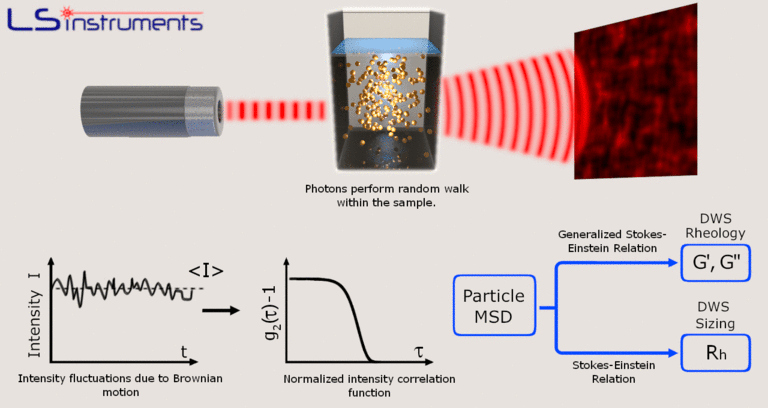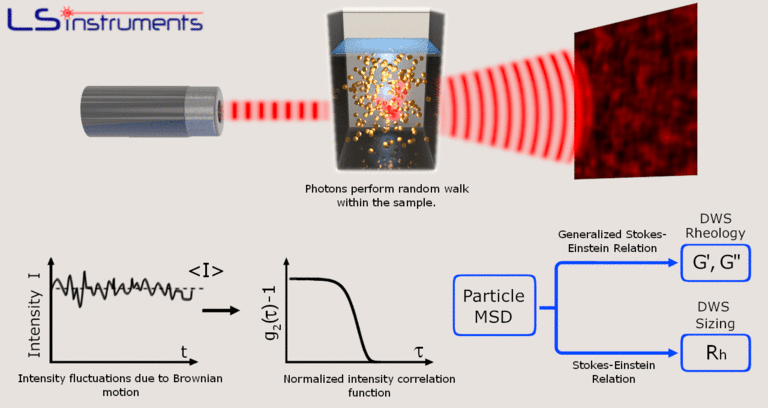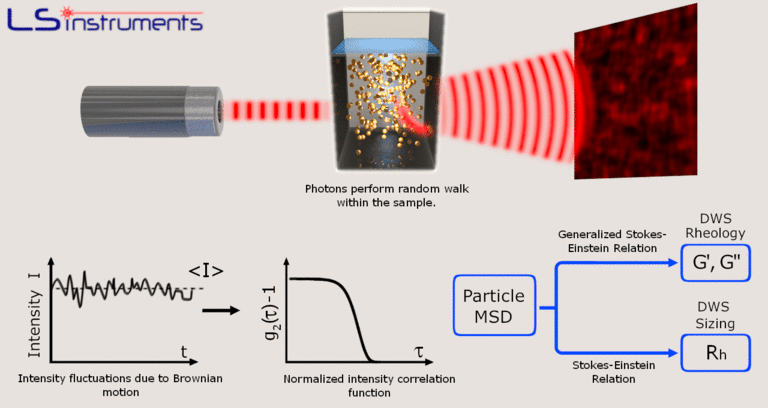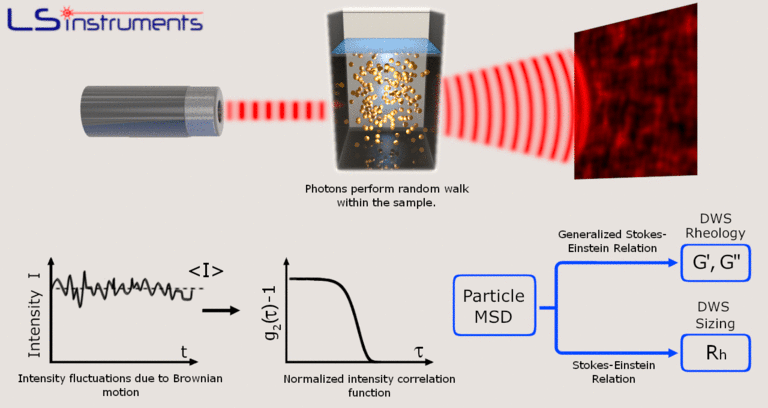
To appreciate why this is the case, two effects faced in concentrated solutions need to be discussed: multiple scattering and mutual diffusion. Although this method is extremely versatile, there are numerous key factors that cannot be ignored when employing light scattering to characterize high-concentration solutions.Īlthough it is feasible to take measurements on high volume-fraction samples without dilution, additional questions arise regarding the meaning of the hydrodynamic size. Sponsored by Brookhaven Instruments Corporation Abstractĭynamic Light Scattering (DLS) is an efficient method of measurement employed for calculating the hydrodynamic size of common nanomaterials consisting of nanoparticles, colloids, polymers and proteins.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
